Activity Description
In less than 15 minutes, you can watch this on-demand, case-based activity from the proceedings of a CE symposium at the 18th Annual Nurse Practitioners in Women’s Health Conference. Learn about:
- The most up-to-date AHA/ACC/TOS, AACE, ENDO, and ANPF obesity guidelines
- Assessing a patient’s readiness to lose weight
- Initiating a weight-loss discussion with your patients
Learning Objectives
Upon completion of this activity, learners should be able to:
- Identify communication strategies to initiate discussions about and increase patient engagement related to weight loss and weight-loss strategies
- Outline patient factors needed to assess readiness for a weight-loss management plan
Faculty

Mary Annette Hess, PhD, FNP-BC, CNS
Assistant Professor/Family Nurse Practitioner
University of Alabama at Birmingham
School of Nursing
Birmingham, AL

Donna H. Ryan, MD, FACP
Professor Emerita
Pennington Biomedical Research Center
Baton Rouge, LA
Faculty Biography
Mary Annette Hess, PhD, FNP-BC, CNS
Mary Annette Hess, PhD, FNP-BC, CNS, is an Assistant Professor at the University of Alabama at Birmingham (UAB) School of Nursing and an Affiliate Member of graduate faculty at the University of Alabama in Huntsville. Dr Hess received her BSN degree at Auburn University at Montgomery in Alabama and her MSN degree in Nursing Education at Troy State University in Alabama. She went on to complete her FNP degree and PhD in Health Education and Health Promotion at UAB.
Dr Hess is an active researcher in the prevention and management of prediabetes, diabetes, dyslipidemia, obesity, and diabetes outcomes and is currently a co-investigator for the NIH-sponsored ENCOURAGE-1 and ENCOURAGE-2 (iAdapt) studies. She has authored or co-authored numerous books, book chapters, and journal articles, and presented her research at several national meetings. She is the Chair for the UAB School of Nursing (SON) Faculty Practice committee and a member of the SON Executive Committee. Dr Hess also serves as an educational member for the National Association of Nurse Practitioners in Women’s Health and Gerontological Advanced Practice Nurses Association, and is on the Board of Directors for Southeastern Diabetes Education Services.
Donna H. Ryan, MD, FACP
Donna H. Ryan, MD, FACP, is Professor Emerita at Pennington Biomedical Research Center in Baton Rouge, Louisiana, where she directed clinical research for 22 years. Her own research includes participation on the teams that developed and executed the DASH (Dietary Approaches to Stop Hypertension), POUNDS Lost, DPP (Diabetes Prevention Program), and Look AHEAD Studies. Dr Ryan was Principal Investigator for 22 years of a Military Nutrition Grant to develop improvements to soldier readiness, nutrition, and health. Dr Ryan’s continuing research interests focus on translation of effective weight management into primary care practices.
Dr Ryan’s scholarly activities include authorship of more than 200 original publications and 45 books, chapters, and reviews, primarily in the field of obesity. She is Associate Editor-in-Chief for Obesity. She served as Co-Chair of the Expert Panel that produced the AHA/ACC/TOS 2013 Guidelines for the Diagnosis and Management of Obesity and was a panel member of the 2015 Endocrine Society Systematic Evidence Review and Guidelines for Medications That Affect Body Weight.
Disclosure Statements
NPWH policy requires all faculty to disclose any affiliation or relationship with a commercial interest that may cause a potential, real, or apparent conflict of interest with the content of a CE program. NPWH does not imply that the affiliation or relationship will affect the content of the CE program. Disclosure provides participants with information that may be important to their evaluation of an activity.
Conflicts of interest were resolved according to NPWH policy prior to development of content.
NPWH policy requires authors to disclose to the participant when presenting information about unlabeled use of any commercial product or device or an investigational use of a drug or device not yet approved for any use.
Faculty
Mary Annette Hess, PhD, FNP-BC, CNS
Dr Hess has no disclosures to be made.
She does not discuss information related to any non-FDA-approved or investigational use of any product/device.
Donna H. Ryan, MD, FACP
Consultant/Advisory Board: Amgen Inc.; Eisai Inc; Janssen Pharmaceuticals, Inc; Novo Nordisk; Pfizer Inc.; Real Appeal; Takeda Pharmaceuticals U.S.A., Inc; Vivus, Inc.
Speaker Bureau: Eisai Inc; Novo Nordisk; Takeda Pharmaceuticals U.S.A., Inc; Vivus, Inc.
Shareholder: Scientific Intake; Tulip Medical, Ltd.
Educational Planning Committee
Spire Learning
Erin Franceschini, MS, Lauren Waters, and Lisa Conklin have nothing to disclose.
National Association of Nurse Practitioners in Women’s Health
Susan Rawlins, MS, WHNP-BC, NP, has nothing to disclose.
Disclaimer
The participating faculty determines the editorial content of the CE activity and the content does not necessarily represent the views of NPWH or Spire Learning. This content has been peer reviewed for validation of clinical content. While every effort has been made to ensure that the information is accurate, clinicians are responsible for evaluating this information in relation to generally accepted standards in their own communities and integrating the information in this activity with those of established recommendations of other authorities, national guidelines, FDA-approved package inserts, and individual patient characteristics.
Off-Label Statement
This educational activity does not contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. The opinions expressed in the educational activity are those of the faculty. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Further, participants should appraise the information presented critically and are encouraged to consult appropriate resources for any product or device mentioned in this program.
Levels of Evidence
Strength of recommendations ratings from the 2013 AHA/ACC/TOS Guideline for the Management of Overweight and Obesity in Adults and the Pharmacological Management of Obesity: An Endocrine Society Clinical Practice Guideline are provided for patient care recommendations made during this activity.
2013 AHA/ACC/TOS Guideline for the Management of Overweight and Obesity in Adults
| NHLBI Grading of the Strength of Recommendations |
| Grade | Strength of Recommendation |
| A | Strong recommendation There is high certainty based on evidence that the net benefit is substantial. |
| B | Moderate recommendation There is moderate certainty based on evidence that the net benefit is moderate to substantial, or there is high certainty that the net benefit is moderate. |
| C | Weak recommendation There is at least moderate certainty based on evidence that there is a small net benefit. |
| D | Recommendation against There is at least moderate certainty based on evidence that there is no net benefit or that risks/harms outweigh benefits. |
| E | Expert opinion (“There is insufficient evidence or evidence is unclear or conflicting, but this is what the Work Group recommends.”) |
| N | No recommendation for or against (“There is insufficient evidence or evidence is unclear or conflicting.”) |
Source: Jensen MD, et al. Obesity. 2014;22:S1-S410.
Pharmacological Management of Obesity: An Endocrine Society Clinical Practice Guideline
Grade 1: Strong recommendation
Grade 2: Weak recommendation
High quality of evidence (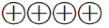 ): Consistent evidence from well performed RCTs or exceptionally strong evidence from unbiased observational studies
): Consistent evidence from well performed RCTs or exceptionally strong evidence from unbiased observational studies
Moderate quality of evidence (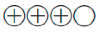 ): Evidence from RCTs with important limitations (inconsistent results, methodological flaws, indirect or imprecise evidence), or unusually strong evidence from unbiased observational studies
): Evidence from RCTs with important limitations (inconsistent results, methodological flaws, indirect or imprecise evidence), or unusually strong evidence from unbiased observational studies
Low quality of evidence (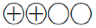 ): Evidence for at least one critical outcome from observational studies, from RCTs with serious flaws, or indirect evidence
): Evidence for at least one critical outcome from observational studies, from RCTs with serious flaws, or indirect evidence
Very low quality of evidence (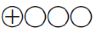 ): Evidence for at least one of the critical outcomes from unsystematic clinical observations or very indirect evidence
): Evidence for at least one of the critical outcomes from unsystematic clinical observations or very indirect evidence
Source: Apovian CM, et al. J Clin Endocrinol Metab. 2015;100:342-362; Swiglo MH, et al. J Clin Endocrinol Metab. 2008;93:666-673.